The arrival of 3D-printed organs isn’t a question of printing speed, but of solving a chain of complex biological and digital hurdles.
- Success depends on creating functional, microscopic blood vessel networks within the tissue.
- Printed organs must then mature in smart bioreactors, a process that is far slower and more complex than the initial printing.
- The digital “blueprint” of your organ, built from your personal genomic data, must be made fundamentally unhackable to ensure safety.
Recommendation: Focus on the phased reality of this technology. Advanced tissues for drug testing are already in use, while complex, fully functional organs for transplant are realistically 10 to 15 years from clinical reality.
The idea of printing a new kidney or heart on demand feels like it’s been pulled directly from science fiction. For anyone on a transplant waiting list, or for those of us fascinated by the future of medicine, it represents a profound hope: the end of organ shortages and the dawn of truly personalized repairs for the human body. We often hear about the promise of using a patient’s own cells to eliminate the risk of rejection, and it seems so logical. The question on everyone’s mind is no longer “if,” but “when?”
The simple answer often thrown around is “in 5 to 15 years.” But as a biomedical engineer working in bioprinting, I can tell you this timeline is not a single finish line. The common perception focuses on the act of printing itself, but that’s just one piece of a much larger puzzle. The real barrier to replacing transplant waiting lists isn’t a single obstacle, but a chain of interconnected biological and digital dependencies that must be solved in sequence. It’s a biological cascade that starts at the microscopic level and ends with the very definition of digital privacy.
The true journey to a 3D-printed heart isn’t about how fast we can print, but about how well we can master the intricate systems that make an organ alive. This involves creating functional blood vessels, patiently guiding cells to mature into complex tissue, and ensuring the digital blueprint of your very own organ is kept absolutely secure. Understanding this chain of challenges is the key to understanding the real, and still incredibly exciting, timeline for this medical revolution.
To fully grasp what it will take to make on-demand organs a reality, this article will walk you through the critical links in this chain of innovation. We will explore each major hurdle and the incredible solutions being developed to overcome them, giving you a realistic and grounded view of the path ahead.
Summary: The Path to On-Demand Organs
- Why Printed Organs Can’t Work If Blood Vessels Don’t Connect?
- How 3D-Printed Liver Tissue Is Replacing Animal Drug Testing?
- 3D Printing vs Cell Maturation: Which Approach to Organ Development Is Faster?
- Will a 3D-Printed Organ from Your Own Cells Ever Be Rejected?
- Will 3D Organ Printing Ever Be Affordable for Patients in Middle-Income Countries?
- When Will AI-Driven Personalised Pills Be Available in High Street Pharmacies?
- How Should Your Genomic Data Be Stored and Shared Safely?
- Will Personalized Medicine Ever Be Affordable for Average Patients?
Why Printed Organs Can’t Work If Blood Vessels Don’t Connect?
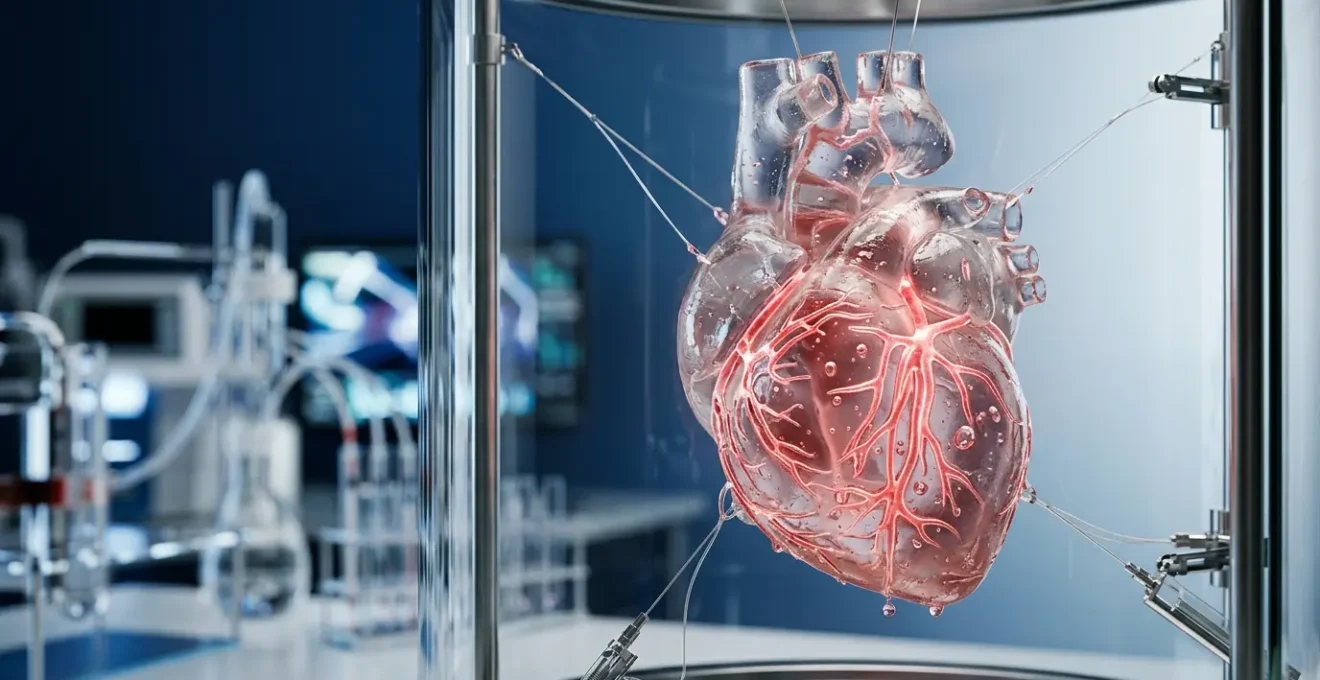
The first and most fundamental hurdle in creating a viable organ is not the organ itself, but its plumbing. You can print a beautiful, anatomically correct structure of cells, but without a network of blood vessels to deliver nutrients and remove waste, it’s just a lifeless sculpture. This is the vascularization challenge. Research confirms that a lack of vasculature limits tissue size and complexity, as cells quickly starve and die. An organ is a living, three-dimensional city; it needs roads and supply lines to function.
To solve this, engineers are developing ingenious techniques that mimic nature’s own processes. One of the most promising is the use of “sacrificial bioink.” In this method, researchers at institutions like CELLINK print a scaffold of the vessel network using a special gel. Once the main organ structure is printed around it, this gel is simply dissolved, leaving behind a perfect, interconnected network of hollow channels. These channels become the organ’s arteries, veins, and capillaries, ready for blood flow. Other methods, like coaxial bioprinting, build the tiny vascular tubes directly during the printing process.

As this image illustrates, the challenge is immense, operating at a microscopic scale to create a lattice of channels with varying diameters. Success in mastering this micro-vascular network is the absolute prerequisite for printing anything larger than a thin sheet of tissue. It is the foundational layer upon which all other progress in complex organ bioprinting is built. Without a solution to this problem, a printed kidney or liver could never survive implantation.
How 3D-Printed Liver Tissue Is Replacing Animal Drug Testing?
While we wait for full organs, a quiet revolution is already happening in pharmacology, thanks to bioprinting. For decades, drug development has relied on animal testing, but this model is notoriously flawed. As the Wyss Institute at Harvard University notes, “Animal models often do not accurately reflect human physiology.” The data backs this up decisively; a stark analysis shows that nearly 90 percent of drug candidates that perform well in animals ultimately fail in human trials, proving to be either ineffective or dangerously toxic.
This is where 3D-printed “organoids” or “organs-on-a-chip” are making their first major impact. Instead of a full organ, scientists are printing small, functional units of human tissue—like miniature liver lobes or lung sacs—using human cells. These organoids provide a far more accurate model for testing a drug’s safety and efficacy. They react just as human tissue would, because they *are* human tissue.
The results are transformative. Take liver toxicity, a major reason for drug failure. A landmark study of 870 “Liver-Chips” demonstrated staggering accuracy. When tested with a range of drugs known to be toxic or non-toxic to the human liver, these chips showed an 87% sensitivity in detecting toxicity and a perfect 100% specificity in confirming non-toxic drugs. This is not just an incremental improvement; it’s a paradigm shift. It means we can get safer, more effective drugs to market faster and at a lower cost, all while dramatically reducing the need for animal testing. This is the first tangible success story of bioprinting, and it’s happening right now.
3D Printing vs Cell Maturation: Which Approach to Organ Development Is Faster?
A common misconception is that the “printing” is the slow part. In reality, the mechanical process of laying down cells can be surprisingly quick. For instance, researchers have shown that a mini-heart took about four hours to print. This speed gives the impression that organs could be produced on an assembly line. However, the printing itself is just the beginning of a much longer and more delicate biological process: maturation.
An organ isn’t just a collection of cells; it’s a sophisticated, functional tissue where cells communicate and work in unison. A freshly printed construct is like a building with all the workers standing in the right rooms but not yet doing their jobs. The process of cell maturation—guiding these cells to form functional tissue—is the true time-consuming step. This is where advanced bioreactors come into play. These devices are essentially artificial wombs for printed organs.

Inside a smart bioreactor, the organ construct is provided with a controlled environment of nutrients, oxygen, and stimuli. As detailed in recent research, these systems can provide pulsing pressure to teach cardiac tissue how to beat or simulate fluid flow to guide kidney cells into forming filtration units. This mechanical and electrical stimulation is what coaxes the cells to mature and organize themselves into a working organ. So while printing takes hours, maturation can take weeks or even months. The real bottleneck isn’t manufacturing speed, but the biological timeline of life itself.
Will a 3D-Printed Organ from Your Own Cells Ever Be Rejected?
One of the greatest promises of bioprinting is the end of organ rejection. Currently, even with a good match, about 10 to 30 percent of transplant patients experience organ rejection and must take immunosuppressant drugs for the rest of their lives. The theory behind bioprinting is simple and elegant: by using a patient’s own cells (autologous cells), the new organ will be a perfect genetic match, making the immune system recognize it as “self” and eliminating the risk of rejection.
For the most part, this theory holds. An organ printed from your own skin or blood cells, reprogrammed into stem cells (known as induced pluripotent stem cells or iPSCs), would be immunologically invisible. However, this powerful technique introduces a different, more subtle risk that must be managed with extreme care. The process of reprogramming cells and growing them at scale carries a small but significant danger.
As experts in the field caution, there is a risk of genetic mutations occurring during the cell culture and differentiation process. This raises a critical safety concern. According to a comprehensive review in PMC on the bioethical issues of bioprinting:
The risk of tumorigenicity is a major problem when using iPSC. To provide safety of iPSC-based therapies, genetic testing of stem cell lines potentially suitable for clinical application has to be performed.
– Bioethical and Legal Issues in 3D Bioprinting Research, PMC review on iPSC safety concerns
This means that while the organ itself won’t be “rejected” by the immune system, there is a non-zero chance that a mutated cell could lead to cancer down the line. Therefore, every batch of cells must undergo rigorous genomic screening before being used. This adds another layer of complexity and cost, but it is an non-negotiable step to ensure patient safety.
Will 3D Organ Printing Ever Be Affordable for Patients in Middle-Income Countries?
With such advanced technology, a major concern is cost. Will 3D-printed organs be a luxury reserved for the wealthy, or can they become a truly global solution? To answer this, we must compare the potential cost of a bioprinted organ not to zero, but to the staggering cost of current treatments for organ failure. The long-term financial burden of managing chronic organ disease is immense.
A stark financial analysis reveals that in the United States, keeping a patient on dialysis costs over a quarter of a million dollars per year, with the average kidney transplant procedure costing over $442,500 in 2020. These are not one-time costs; they represent a lifelong drain on healthcare systems and family finances. When viewed through this lens, the upfront cost of bioprinting a new, permanent organ starts to look not just competitive, but economically advantageous.
Furthermore, the cost of the technology itself is falling faster than many realize. While the most advanced research printers cost millions, market research indicates that professional-grade bioprinters… are available on the commercial market for as low as $25,000. As with all technologies, from computers to gene sequencing, costs will continue to decline due to economies of scale and process improvements. The initial investment in establishing bioprinting facilities could be high, but the long-term saving from curing—rather than managing—chronic disease presents a powerful argument for its adoption, even in middle-income countries looking to build more sustainable healthcare systems.
When Will AI-Driven Personalised Pills Be Available in High Street Pharmacies?
While the title mentions personalized pills, the underlying technology—Artificial Intelligence—is a critical accelerator in the quest for personalized organs. AI is not just a buzzword here; it is an essential tool for overcoming the complexity and time constraints of bioprinting. Its impact is felt across the entire development chain, from initial testing to final maturation.
We’ve already seen how organs-on-a-chip are revolutionizing drug testing. AI takes this a step further. By using patient-derived cells on these chips, AI algorithms can model patient-specific responses to various drug cocktails, creating a “digital twin” for risk-free experimentation. This allows doctors to test and optimize personalized treatments on a chip before ever administering them to the patient, dramatically improving safety and efficacy for everything from chemotherapy to organ transplant medications.
AI’s role becomes even more critical during the slow maturation phase inside a bioreactor. As experts in the field have noted, AI is the key to speeding up this biological timeline. In an article from Nature Scientific Reports, researchers explain:
AI algorithms can monitor thousands of data points from sensors within the bioreactor in real-time and dynamically adjust the environment (oxygen, nutrients, stimuli) to optimize and accelerate the organ’s development.
– Advanced Bioprinting Research, Nature Scientific Reports on bioreactor systems
In essence, AI acts as the master gardener for the developing organ. It can make micro-adjustments far faster and more accurately than any human could, optimizing the growth conditions to achieve functional maturity in the shortest possible time. This synergy between biology and AI is what will ultimately make the “5 to 15 year” timeline a reality.
How Should Your Genomic Data Be Stored and Shared Safely?
As we move closer to printing personalized organs, we encounter a challenge that is not biological, but digital: data security. Creating a custom organ requires an incredibly detailed digital file, a true “organ blueprint.” This file combines a patient’s complete genomic data with anatomical information from MRI or CT scans. It is, without exaggeration, one of the most sensitive and personal pieces of information that could ever exist.
The security implications are profound. As a comprehensive review highlights, this organ blueprint becomes a critical point of vulnerability. Unlike a mass-produced drug, a custom-printed organ cannot be tested on anyone else before implantation. The digital file is the single source of truth. The risk of that file being stolen, tampered with, or even held for ransom is a serious ethical and security nightmare. Who owns this data? How is it encrypted? How do we prevent unauthorized replication? These questions must be answered before the technology can be widely deployed.

Protecting this data will require a new paradigm of cybersecurity, likely involving blockchain for data integrity and advanced biometric encryption. The file for your new heart cannot be stored like a simple medical record; it must be treated with the same level of security as a nation’s most sensitive secrets. This digital challenge is the final, and perhaps least understood, link in the chain of dependencies. Without a solution, the entire promise of personalized organs is at risk.
Action Plan: Key Points for Future Organ Blueprint Security
- Data Ownership: Clarify who has ultimate legal ownership of your genomic blueprint—you, the hospital, or the bioprinting company.
- Encryption Standards: Inquire about the level of encryption used, demanding end-to-end encryption and multi-factor authentication for any access.
- Access Logs: Ensure that any system storing your blueprint uses an immutable ledger (like blockchain) to track every single time the file is accessed or modified.
- Deletion Protocols: Understand the protocols for the secure and permanent deletion of your data after the organ has been printed and successfully transplanted.
- Regulatory Compliance: Verify that the storage and handling of your data comply with the highest national and international data privacy regulations (like GDPR or HIPAA).
Key Takeaways
- The primary biological hurdle is vascularization; organs need a fully integrated micro-vessel network for nutrient and waste transport to survive.
- The main time constraint is not printing but maturation, a weeks-long biological process in smart bioreactors where cells are stimulated to form functional tissue.
- A critical and often overlooked challenge is digital security; the “organ blueprint” containing a patient’s genomic data must be protected with state-of-the-art encryption to prevent theft or tampering.
Will Personalized Medicine Ever Be Affordable for Average Patients?
After navigating the chain of biological, engineering, and digital challenges, we return to the ultimate questions: when will this all be available, and can an average person afford it? The synthesis of all these factors gives us a realistic, phased timeline. Simpler, avascular tissues like skin and cartilage are already in clinical use. More complex tissues for drug testing are mainstream. But for full, internal organs with complex vascular networks, a pragmatic forecast from experts suggests it will be 15 to 20 years for their availability in clinical trials.
Regarding affordability, the perspective of leading figures in the field is illuminating. The high upfront cost must be weighed against the immense, recurring costs of managing chronic illness. As Dr. Anthony Atala, a pioneer at the Wake Forest Institute for Regenerative Medicine, powerfully argues:
The costs associated with organ failures are very high. Just to keep a patient on dialysis is over a quarter of a million dollars per year. So, it’s a lot cheaper to create an organ that you can implant into the patient.
– Dr. Anthony Atala, Wake Forest Institute for Regenerative Medicine
This is the core economic argument for personalized medicine. While the initial price tag will be high, the cost will decline over time. More importantly, it represents a shift from a model of endless, expensive management to one of a permanent, curative solution. The ultimate affordability of a printed organ lies in its ability to provide a lifetime of health, ending the crushing financial and personal burden of chronic organ failure. It is an investment, not just an expense.
The journey towards on-demand, 3D-printed organs is one of the most exciting endeavors in modern medicine. By understanding the real challenges and the brilliant solutions being developed, we can maintain a sense of hopeful realism. The next step for us all is to stay informed, support medical research, and advocate for the thoughtful and ethical deployment of these life-changing technologies.