
The extreme cost of a surgical instrument has almost nothing to do with the metal and everything to do with the legally-defensible guarantee of absolute sterility.
- A hardware store tool is clean; a surgical instrument meets a “Sterility Assurance Level” where the chance of a single living microbe is one in a million.
- The price includes a lifetime of tracking, testing, and documentation to prove its sterility for every single procedure.
Recommendation: Instead of questioning the price of the tool, question the robustness of the sterilisation and tracking system. That is where true patient safety lies.
It’s a question that surfaces in the mind of any observant patient or cost-conscious hospital administrator: why does a simple-looking steel clamp or scalpel carry a price tag that could buy a used car? You can walk into a hardware store and buy a set of precision tools, made of high-grade steel, for a tiny fraction of the cost. They look similar. They feel similar. So where does the immense, four-figure difference in value come from?
The common answers – that surgical instruments are made of “special stainless steel” or require “precision manufacturing” – are true, but they are profoundly incomplete. They miss the fundamental point. The cost isn’t just for the physical object you can hold. It’s for an invisible, complex, and non-negotiable system of absolute certainty. You aren’t buying a tool; you are buying a guarantee. A guarantee that the instrument entering a patient’s body is not just clean, not just disinfected, but sterile to a degree that is statistically almost perfect.
This article is not about the metal. It’s about the process. We will dismantle the true cost of a surgical instrument by exposing the rigorous, uncompromising, and legally mandated world of sterilisation, traceability, and quality assurance. You will learn why boiling water is dangerously inadequate, how a hospital can prove an instrument was sterile years after your surgery, and why the human element is the weakest link in a chain that must be unbreakable. The difference between a £5 craft knife and a £5,000 scalpel is the difference between a calculated risk and a verifiable certainty. In an operating theatre, there is no room for anything but the latter.
To fully grasp the chasm between a simple tool and a medical-grade instrument, we will explore the critical processes that define surgical safety. The following sections break down the science, technology, and protocols that are the true source of an instrument’s cost and, more importantly, its ability to protect a patient.
Summary: Why a £5,000 Price Tag on a Scalpel is a Guarantee of Safety
- Why “Sterile” Instruments Undergo 121°C Steam for 15 Minutes Specifically?
- How Can a Hospital Prove an Instrument Was Sterile During Your Operation?
- Single-Use vs Reusable: Is Disposable Really Better for Safety?
- The Human Error That Compromises Sterile Field During 2-Hour Surgery
- How Many Times Can a £3,000 Instrument Be Reused Safely?
- Why Boiling Water Is Not Enough to Kill Hepatitis B on Tools?
- NHS Safety Protocols vs Private Clinic Rules: Are You More Protected?
- Why Your Dentist Uses an Autoclave for Every Patient?
Why “Sterile” Instruments Undergo 121°C Steam for 15 Minutes Specifically?
The parameters of 121°C for 15 minutes are not arbitrary; they are a clinical standard derived from killing the toughest life forms on Earth. This isn’t about killing common bacteria. It’s about annihilating highly resistant bacterial spores. To be considered sterile for surgical use, industry standards mandate that surgical instruments achieve a Sterility Assurance Level of 10^-6. This means there is a one-in-a-million probability of a single non-sterile item in a sterilised batch. A hardware store tool, by contrast, has a near 100% probability of being non-sterile.
To validate this process, we use a “biological indicator.” This involves placing a vial of one of the most heat-resistant organisms known, Geobacillus stearothermophilus, inside the autoclave with the instruments. The image below shows what these indicators look like. If the autoclave cycle successfully kills these super-resilient spores, we can be confident it has destroyed everything else.

This spore test is the ultimate proof. As sterilization standards organizations confirm, the entire process is benchmarked against this specific threat. As they state, “Geobacillus stearothermophilus spores are highly resistant to heat, and their destruction at 121°C confirms the effectiveness of the autoclave.” This scientific validation is a core part of the instrument’s cost—it’s the price of certainty, proven by challenging the process with the strongest possible adversary.
How Can a Hospital Prove an Instrument Was Sterile During Your Operation?
A hospital proves sterility not with a promise, but with a data trail. In a modern operating theatre, every critical instrument is part of a comprehensive tracking system, creating an unbroken chain of custody from the patient, through the decontamination and sterilisation process, and back to storage. This isn’t just good practice; it’s a legal and regulatory necessity. If a post-operative infection occurs, the hospital must be able to produce records proving that every instrument used was successfully sterilised.
This is achieved through a combination of physical logs, chemical indicators on the packaging, and increasingly, Radio-Frequency Identification (RFID) tags. Each instrument or tray is scanned at every stage: disassembly, cleaning, inspection, packaging, sterilisation, and use. The autoclave’s own records—showing it reached the correct temperature and pressure for the required duration—are linked to that specific instrument’s cycle. This is such a critical area of investment that the surgical instrument tracking system market is projected to grow from $203 million to $406 million between 2021 and 2026. This investment is not in the steel; it’s in the data that ensures the steel is safe.
Your Audit Checklist: Verifying the Chain of Sterility
- Unique Identification: Does every instrument or tray have a unique barcode or RFID tag for individual tracking?
- Cycle Data Log: Can you pull the specific autoclave cycle data (time, temperature, pressure) for the exact time an instrument was processed?
- Biological Indicator Results: Are the results of the spore tests (the ultimate proof of killing power) logged and linked to the autoclave’s cycles?
- Chemical Indicator Check: Does the packaging for each instrument show visual proof (e.g., a colour-changing strip) that it has been exposed to the sterilisation process?
- Technician Accountability: Is the ID of the sterile processing technician who packed and inspected the instrument logged in the system?
This meticulous, and expensive, record-keeping is the hospital’s defence and the patient’s protection. It’s the tangible proof that stands up in court and, more importantly, prevents infections before they start.
Single-Use vs Reusable: Is Disposable Really Better for Safety?
The rise of single-use, disposable instruments was driven by a seemingly simple premise: a brand-new, sterile-packaged item eliminates any risk of cross-contamination from improper reprocessing. For certain complex devices with hard-to-clean lumens or intricate mechanisms, this logic is sound. It removes the variables of human error in cleaning and the potential for biofilm build-up. From a purely risk-averse standpoint, disposable seems like the ultimate safety net.
However, this is not a one-sided argument. The assumption that “disposable is always better” is being challenged on multiple fronts. Firstly, the environmental impact is staggering. The healthcare industry is a massive contributor to landfill waste and carbon emissions. Research is now showing that for many common instruments, the lifecycle of a reusable item is far more sustainable. In fact, studies indicate that switching from disposable to reusable surgical instruments can achieve a 40-66% reduction in CO2 emissions.
Secondly, there is the question of quality. A £3,000 reusable retractor is engineered for performance and durability over hundreds of cycles. A single-use plastic equivalent may not provide the same rigidity, feel, or reliability in the surgeon’s hand. The decision is therefore a complex balance of risk, cost, surgeon preference, and environmental responsibility. It’s not a simple case of one being universally “better.” The critical factor for reusables is the absolute robustness of the cleaning and sterilisation process. If that system is flawless, the safety is equivalent, and the benefits in cost and sustainability are significant.
The Human Error That Compromises Sterile Field During 2-Hour Surgery
The sterilisation process can be perfect, the instruments flawlessly tracked, yet safety can be compromised in a split second within the operating theatre itself. The single greatest variable is human error. Maintaining the sterile field—the designated area around the patient that is considered free of microorganisms—is an act of constant, disciplined vigilance. Every member of the surgical team is trained to treat the boundaries of this field, typically marked by blue drapes, as absolute. Nothing unsterile can cross that line.
The image below illustrates this concept, showing the carefully arranged instruments and drapes that define the zone of asepsis. A single accidental touch of a mask, an ungloved hand reaching over the field, or a non-sterile object falling onto the tray can invalidate the entire setup, forcing a costly and time-consuming reset.
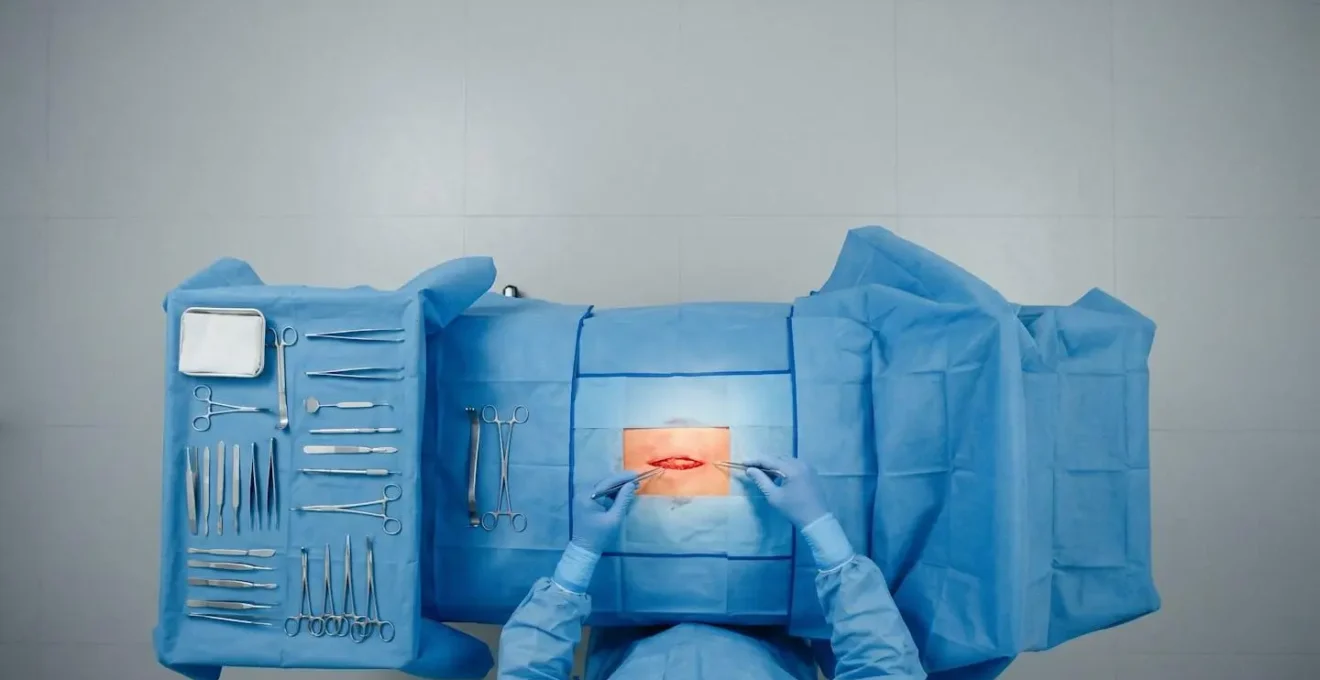
Another critical area for human error is instrument and sponge counts. The protocol of manually counting every item before, during, and after a procedure is a foundational safety check to prevent retained surgical items. Yet, it is fallible. The pressure and focus of a long, complex surgery can lead to mistakes. Shockingly, research shows that in up to 88% of surgical retention cases, the team had recorded erroneously correct manual counts. This demonstrates that even diligent human processes can fail, which is why technology like barcoding or RFID-tagged sponges are being implemented as a backup system. The cost of these technologies is a direct investment in mitigating the unavoidable risk of human fallibility.
How Many Times Can a £3,000 Instrument Be Reused Safely?
The lifespan of a high-end surgical instrument is not measured in years, but in the number of reprocessing cycles it can endure while maintaining its functional integrity and safety. A £3,000 instrument is an asset designed for longevity. The manufacturer provides specific guidelines based on extensive testing, defining how many times the instrument can go through the harsh process of cleaning with enzymatic detergents and sterilisation with high-pressure, high-temperature steam before it must be retired.
For a standard stainless steel instrument like a retractor or forceps, the number of cycles can be in the hundreds or even thousands, provided it passes a rigorous inspection each time. Technicians check for corrosion, cracking, stiffness of joints, and sharpness of blades. Any sign of wear and tear that could either compromise its function or its ability to be cleaned effectively means it is immediately removed from service. This is another reason for the high initial cost: the materials and construction are chosen to withstand this punishing cycle repeatedly without degradation.
The tracking technology used to monitor this is also built for this environment. It’s not enough to simply track an instrument; the tracking mechanism itself must survive. As an example of this durability, modern RFID tracking tags attached to surgical instruments can withstand up to 2,500 sterilisation cycles. This ensures the data connection remains intact for the entire usable life of the instrument. The answer to “how many times” is therefore determined by the manufacturer’s validated limit and the results of a meticulous inspection after every single use. The system is designed so that an instrument is never used beyond its proven safe lifespan.
Why Boiling Water Is Not Enough to Kill Hepatitis B on Tools?
The idea that boiling water (100°C) is a sufficient sterilant is a dangerous misconception. While boiling can kill many common bacteria and viruses, it is completely inadequate for a surgical environment for two critical reasons: it cannot kill heat-resistant spores, and it cannot reliably inactivate certain hardy viruses like Hepatitis B or infectious proteins like prions. This is a matter of pure physics and biology. True sterilisation requires a higher temperature that water simply cannot reach at standard atmospheric pressure.
This is where the autoclave comes in. By increasing the pressure inside the chamber to about 15 psi (pounds per square inch) above atmospheric pressure, it allows water to remain liquid up to a much higher temperature. It is this combination that is key. As Sterilization Science Standards explain, “The pressure itself is not the sterilizing agent; it is simply the tool that allows water to be heated to 121°C.” This 21-degree increase is the difference between disinfection and sterilisation. The sterilization temperatures of 121°C achieved via 15 psi pressure provide the thermal energy required to denature the proteins of even the most resilient microorganisms and viruses, ensuring their destruction.
At 121°C, the thermal death time for most microorganisms, including heat-resistant bacterial spores, is significantly reduced. The pressure itself is not the sterilizing agent; it is simply the tool that allows water to be heated to 121°C.
– Sterilization Science Standards, Why Is 121 the Standard Autoclave Temperature for Sterilization?
A tool from a hardware store might survive being boiled, but it wouldn’t be considered safe for surgery. The cost of a surgical instrument is intrinsically tied to the fact that it is engineered to withstand and be rendered sterile by the far more aggressive conditions of an autoclave, a process that guarantees the elimination of threats that boiling water leaves behind.
NHS Safety Protocols vs Private Clinic Rules: Are You More Protected?
It’s a common assumption that the core safety protocols for instrument sterilisation are universal, and to a large extent, they are. Regulatory bodies like the Care Quality Commission (CQC) in the UK mandate a baseline standard of care that applies to both the NHS and private clinics. The principles of cleaning, disinfection, and sterilisation using validated autoclave cycles are non-negotiable for any registered healthcare provider. On paper, the rules are the same for everyone.
However, the true level of protection for a patient is often defined not by the written rules, but by the culture and resources dedicated to upholding them. A large NHS trust may have a dedicated, state-of-the-art Sterile Services Department (SSD) with highly trained technicians and significant capital for the latest tracking technology. A smaller private clinic might outsource its sterilisation or have a smaller-scale in-house operation. This isn’t to say one is inherently safer, but the scale and resources can differ.
The ultimate determinant of safety is not the name over the door, but the organisation’s commitment to a “culture of safety.”
A hospital’s true level of protection is defined by its investment in training for sterile processing staff, its non-punitive error reporting systems, and empowering every employee to halt a procedure if safety is in doubt.
– Healthcare Safety Culture Analysis, A Day in the Life of a Surgical Instrument: The Cycle of Sterilization
Therefore, a patient is most protected in an environment—be it NHS or private—that invests heavily in its systems, trains its people relentlessly, and fosters a culture where raising a safety concern is not just welcomed, but required. The rules might be the same, but the rigorousness of the implementation is what creates the margin of safety.
Key Takeaways
- The cost of a surgical instrument is not for the steel, but for the verifiable, one-in-a-million guarantee of sterility (Sterility Assurance Level).
- This guarantee is proven through a documented chain of custody, using tracking systems and biological spore tests for every sterilisation cycle.
- The choice between reusable and single-use is a complex trade-off of risk, quality, cost, and environmental impact, not a simple “one is safer” argument.
Why Your Dentist Uses an Autoclave for Every Patient?
The dental clinic provides a perfect, small-scale illustration of these non-negotiable principles. While the procedures may be less invasive than major surgery, the risk of transmitting blood-borne pathogens like Hepatitis B, Hepatitis C, or HIV is just as real. Every instrument that comes into contact with a patient’s mouth is considered contaminated and must undergo the exact same calibre of reprocessing as a surgical tool in a hospital.
Your dentist uses an autoclave for the same reason a surgeon does: it is the only verifiable method to achieve sterilisation, not just disinfection. The volume of work is significant; guidelines from the WHO suggest that each sterile processing technician processes 1,500-2,000 instrument sets per year, a standard that applies to the scale of work in dental practices as well. Each set of probes, mirrors, and scalers is packaged in a special pouch with a chemical indicator that changes colour, providing you with immediate visual proof that the pouch has been through the autoclave cycle.
This commitment is the bedrock of patient trust and public health. Research from institutions like Duke University has explored using advanced tracking in these settings to ensure compliance and efficiency. As their research on instrument tracking notes, the process is systematic:
Each tagged instrument was scanned into a database pairing tag ID numbers to instrument information. Tags and instrument tape are autoclave-compatible, and tagged instruments were processed through the conventional instrument sterilization cycle.
– Duke University Medical Research Team, Measuring intraoperative surgical instrument use with radio-frequency identification
The next time you see your dentist open that sealed pouch, you are not just witnessing good hygiene. You are witnessing the end point of a rigorous, costly, and essential process that is identical in principle to the one that protects you in a major operating theatre. It is the minimum standard of care, and there is no room for compromise.
To ensure patient safety, the entire system must be viewed as a single, integrated process. The value of a £5,000 instrument lies not in its material, but in its proven ability to withstand and be verified by this uncompromising system, protecting both the patient and the institution from the catastrophic consequences of a single failure. Assess and demand this level of systemic integrity for your care.