The short answer: an AI app can’t replace a dermatologist, but it can be a powerful tool to get you to one faster.
- AI excels at rapid pattern recognition for common conditions but often fails with rare or unusual presentations due to “data blind spots.”
- Consumer symptom-checker apps have a dangerously wide accuracy range (from 23% to 70%) and can amplify health anxiety.
Recommendation: Use AI tools not for a definitive diagnosis, but as a sophisticated triage system to justify an urgent medical consultation and to have a more informed conversation with your doctor.
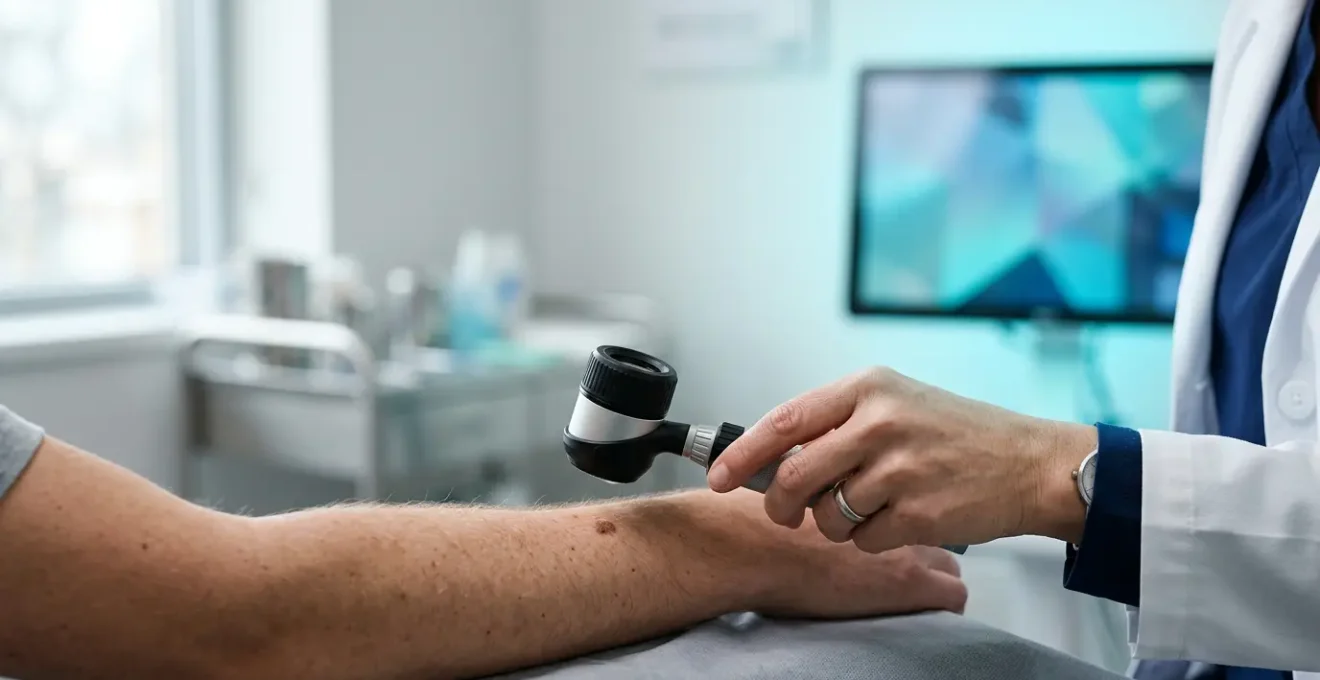
That feeling—a pit in your stomach when you notice a new or changing mole. Your first thought is to see a doctor, but then reality hits: the long wait for a GP appointment, followed by an even longer wait for a dermatology referral. In this gap of anxiety, technology offers a tempting solution: an app that promises to check your skin with a simple photo. The question on everyone’s mind is no longer just “Is this serious?” but “Can an app give me an answer right now?”
For years, the standard advice has been to avoid “Dr. Google” and wait for a professional opinion. But this advice ignores the very real emotional toll and potential clinical risk of waiting. The landscape is changing. Artificial intelligence is no longer a futuristic concept; it’s a diagnostic assistant already at work in NHS hospitals. This isn’t about replacing doctors. It’s about augmenting their abilities with incredible speed and data-processing power.
The crucial shift in thinking is moving from “AI or doctor?” to “AI *then* doctor.” The real power of these tools isn’t to give you a final, definitive diagnosis at home. It’s to act as an intelligent, data-driven reason to either seek immediate professional help or gain peace of mind. This guide will cut through the hype and fear. We will explore where AI diagnostics excel, where their critical blind spots lie, and how you can use this technology as an empowered patient to navigate the healthcare system more effectively.
To understand this new reality, this article breaks down the capabilities and limitations of diagnostic AI. We’ll explore its use across different medical fields, from spotting rare diseases to analysing heart scans, and provide a clear framework for when to trust the algorithm and when to demand a human expert.
Summary: Your Guide to AI in Medical Diagnostics
- Why AI Diagnostics Still Miss 1 in 10 Rare Conditions?
- How to Get an AI-Assisted Heart Scan on the NHS This Year?
- Radiologist vs AI Algorithm: Who Spots Pneumonia First?
- The Danger of Relying Solely on Health Apps for Symptom Checking
- How to Prepare Your Medical History for an AI Analysis Tool?
- Why Commercial DNA Tests Are Not Accepted by NHS Doctors?
- What Does ‘Incidental Lung Nodule’ Mean and Should You Worry?
- Should You Pay £200 for a Private DNA Health Test?
Why AI Diagnostics Still Miss 1 in 10 Rare Conditions?
Artificial intelligence learns from data. Its incredible ability to spot patterns in thousands of images of common skin cancers is revolutionary. But what happens when it encounters something it has never seen before? This is AI’s fundamental weakness: data blind spots. An algorithm trained primarily on data from light-skinned populations may struggle to accurately assess a lesion on dark skin. Similarly, a rare disease, by definition, has very little data available for training.
While AI is powerful, it lacks a doctor’s intuition and ability to connect disparate symptoms. As Dr. Vivian Schmolly noted in a UCLA Health study, the consequences of these gaps are profound. She explains the challenge for human physicians:
The diagnostic delay is roughly 10 to 15 years for these diseases because physicians don’t see them very often. And while waiting for diagnosis, the disease can progress and cause irreversible damage.
– Dr. Vivian Schmolly, UCLA Health study on zebraMD AI tool for rare diseases
Even so, AI is making remarkable progress. For instance, recent research from UCLA and UCSF demonstrates that a tool named zebraMD achieved an accuracy of 89-93% in identifying rare genetic disorders from patient photos. This doesn’t eliminate the need for a specialist, but it dramatically shortens the diagnostic odyssey, turning a decade of uncertainty into a rapid, targeted referral. The AI acts as a powerful triage tool, flagging possibilities that a GP might not consider.
How to Get an AI-Assisted Heart Scan on the NHS This Year?
Beyond theoretical discussions, AI is already an active partner in NHS diagnostics. A prime example is in cardiology, traditionally an area reliant on invasive and time-consuming procedures. For patients with suspected coronary heart disease, the standard path often involves an angiogram, which carries risks and requires hospital time. Today, a non-invasive alternative is being rolled out across England.
The technology, called HeartFlow, uses AI to analyse a standard CT scan of the heart. It creates a personalised, 3D digital model of the coronary arteries and assesses the impact of any blockages on blood flow. This gives cardiologists a wealth of information without an invasive procedure. According to NHS England data, this technology is already in use in at least 56 NHS hospitals, having helped over 6,800 patients in early 2024 and significantly reducing the need for angiograms.
For a patient concerned about heart symptoms, knowing this technology exists is empowering. It changes the conversation with your GP from a general concern to a specific inquiry. Here are the steps you can take:
- Consult your GP about symptoms of suspected coronary heart disease and request a referral for a cardiac assessment.
- Ask your GP or cardiologist specifically about the availability of HeartFlow Analysis at your local or referred hospital.
- Understand that this AI-driven tool converts a standard CT scan into a detailed 3D map, potentially saving you from an invasive angiogram.
- You can also inquire about enrollment in clinical trials for even newer AI cardiac MRI tools, which can analyse heart function in seconds and are available at pioneering centres like UCL Hospital and Barts Heart Centre.
This shows AI is not just a future promise but a present-day reality that patients can proactively access. It serves as a tool for both the doctor and the patient, enabling faster, safer, and more accurate diagnoses.
Radiologist vs AI Algorithm: Who Spots Pneumonia First?
In the field of radiology, the volume of images a single radiologist must review is immense, leading to fatigue and the potential for error. This is where AI shines, not as a replacement, but as an tireless, vigilant assistant. The “centaur model,” where a human expert is paired with an AI, is becoming the gold standard. The AI performs a rapid first-pass analysis, flagging potential abnormalities for the radiologist to review and confirm.
This synergy is particularly effective in detecting conditions like pneumonia from chest X-rays. An AI can analyse an image in seconds, spotting subtle opacities that might be missed on a quick human read. The evidence for this is strong; a meta-analysis of 15 studies found that AI models achieved an average sensitivity of 88% and specificity of 90% for pneumonia detection. This means they are very good at both correctly identifying the disease and correctly ruling it out.
This image represents the modern diagnostic workflow. The AI isn’t making the final call; it is augmenting the radiologist’s expertise. It prioritises suspicious cases, reduces turnaround time from hours to minutes, and acts as a safety net. For the patient, this means a faster diagnosis and quicker start to treatment, which is critical in time-sensitive illnesses like pneumonia. The final diagnosis remains a human-in-the-loop decision, combining the AI’s speed with the radiologist’s contextual knowledge and experience.
The Danger of Relying Solely on Health Apps for Symptom Checking
While clinical AI is transforming hospital diagnostics, the world of direct-to-consumer health apps is a different story. The promise is alluring: get an instant risk assessment of your symptoms from your phone. However, the reality is fraught with risk, primarily due to a wild variance in quality and a dangerous psychological feedback loop.
The diagnostic accuracy of these apps is alarmingly inconsistent. A clinical vignettes study comparing eight popular symptom apps revealed a diagnostic accuracy range from as low as 23.5% to a high of 70.5%. Relying on an app with a one-in-four chance of being correct is a high-stakes gamble. The greater danger, however, lies in how these apps interact with user anxiety. For someone already worried about a health issue, an inaccurate or ambiguous result can be fuel for a fire of hypochondria.
This isn’t just speculation; it’s a documented phenomenon. As researchers from the University of Tübingen found, the user base of these apps is particularly vulnerable.
Over half of the symptom checker app users had clinically relevant hypochondria. This level of anxiety may affect a patient’s ability to adequately handle action recommendations and symptom classifications, making these users susceptible to the negative effects of app use.
– Researchers from University of Tübingen, BMC Medical Informatics and Decision Making
An app might incorrectly flag a benign mole as high-risk, causing unnecessary panic. Conversely, and more dangerously, it might dismiss a potentially malignant melanoma as “low-risk,” providing false reassurance and delaying a crucial doctor’s visit. The only safe way to use these apps is to treat any result—positive or negative—as a prompt to seek professional medical advice, not as a substitute for it.
How to Prepare Your Medical History for an AI Analysis Tool?
The principle of “garbage in, garbage out” is paramount in artificial intelligence. An AI diagnostic tool, whether used by you or your doctor, is only as good as the data it receives. A single photo of a mole lacks crucial context: Has it changed? Do you have a family history of skin cancer? Are you on any new medications? A doctor synthesises this information automatically, but an AI needs it explicitly.
Therefore, one of the most powerful things you can do as a patient is to become the curator of your own health data. Transforming your messy, unstructured medical history into a clean, structured health record makes it more useful for both human doctors and AI tools. This involves documenting symptoms with dates, tracking medications, and noting major life events. A well-organized record can reveal patterns that neither you nor your doctor might have noticed.
Taking control of your data prepares you for a future where you can provide an AI with a comprehensive picture, leading to a much more accurate and personalised analysis. It empowers you to have more productive conversations with your healthcare providers.
Your 5-Point Audit for AI-Ready Health Records
- Points of Contact: List all sources of your health data—your GP, any specialists, hospital visits, and data from health apps or wearables (e.g., heart rate, sleep patterns).
- Collect: Gather your existing records. Request digital or paper copies of lab results, imaging reports, and consultation notes. Inventory all current and past prescriptions.
- Coherence: Cross-reference the data for consistency. Does a symptom log you kept align with a diagnosis date? Are there any unexplained gaps or contradictory entries?
- Mémorability & Emotion: Identify the key turning points in your health history versus routine data. Highlight major diagnoses, surgeries, or allergic reactions. This creates a narrative, not just a list.
- Plan for Integration: Consolidate everything into a single, portable digital format (like a secure document or a dedicated personal health record app). This becomes your master file to share with new doctors or future AI tools.
Why Commercial DNA Tests Are Not Accepted by NHS Doctors?
The rise of direct-to-consumer (DTC) DNA tests, offering insights into everything from ancestry to health risks for a couple of hundred pounds, is another area where technology and medicine collide. Many people take these tests and bring the results to their GP, expecting it to be integrated into their NHS care, only to be met with skepticism. The reason for this is a critical distinction between “interesting information” and clinically validated data.
NHS genetic testing is a highly regulated diagnostic process. It uses specific, targeted tests to answer a clear clinical question (e.g., “Does this patient have the BRCA1 gene mutation?”). The labs are accredited, the results are interpreted by clinical geneticists, and there are clear care pathways based on the outcome. This rigorous process is essential because the stakes are so high.
Commercial tests, on the other hand, often operate in a different sphere. They use a technology called genotyping, which scans for common genetic markers across your genome. While this is great for ancestry, its application to complex diseases is often probabilistic and not definitive. The tests are not subject to the same stringent validation as medical devices, a point we’ll explore further. The need for better diagnostics is clear, as research from Baylor Genetics reveals that even with advanced clinical tools, the diagnostic rate for rare genetic disorders is only around 30%, with an average delay of six years. However, the current generation of DTC tests is not the solution the NHS can rely on.
What Does ‘Incidental Lung Nodule’ Mean and Should You Worry?
An “incidental finding” is something a doctor discovers by chance while looking for something else. With the increasing power of imaging technology and AI, this is happening more often. A common example is an “incidental lung nodule” found on a CT scan ordered for an unrelated reason. The term itself can sound alarming, but in most cases, these nodules are benign (non-cancerous) scars from past infections.
Here again, AI is a double-edged sword. It’s incredibly good at spotting these tiny nodules, things the human eye might easily miss. A meta-analysis shows AI achieves 72% sensitivity in detecting lung nodules on chest X-rays, often flagging them for further investigation. This can lead to earlier detection of lung cancer in a small number of cases. However, it also leads to more follow-up scans and anxiety for the vast majority of people whose nodules are harmless.
The key is not to panic, but to understand the language of risk that radiologists use. Your follow-up will depend on the nodule’s size, shape, and density. This table helps translate the terminology you might see on a radiology report.
| Radiology Term | Size Threshold | Risk Implication | Typical Next Step |
|---|---|---|---|
| Micronodule | <3mm | Very low risk, often benign | No follow-up usually needed |
| Small nodule | 3-8mm | Low-moderate risk | Follow-up CT in 6-12 months |
| Spiculated nodule | Variable | Higher risk (irregular borders) | Biopsy or PET scan consideration |
| Ground-glass opacity | Variable | May indicate early adenocarcinoma | Close monitoring, potential biopsy |
| Calcified nodule | Variable | Typically benign | Usually no action required |
An AI might flag the nodule, but a human radiologist assesses these features to determine the actual risk and recommend the appropriate next step. Once again, it is the human-in-the-loop who makes the final judgement call.
Key takeaways
- AI’s primary strength in medicine is as a triage and pattern-recognition tool, not as a final diagnostician; it excels at speed and scale for common conditions.
- The accuracy of direct-to-consumer health apps is dangerously inconsistent. They should be used only as a prompt for a professional consultation, not as a replacement.
- For AI to provide meaningful insights, it requires high-quality, structured data. You can significantly improve your care by becoming the curator of your own medical records.
Should You Pay £200 for a Private DNA Health Test?
So, we return to the question: is it worth spending £200 on a private DNA health test? The answer is nuanced: it depends entirely on your expectations. If you are seeking entertainment, ancestry information, or non-medical wellness tips (e.g., “you may be a fast caffeine metaboliser”), these tests can be fascinating. However, if you are seeking a medical diagnosis or a definitive risk assessment for a serious disease, you must proceed with extreme caution.
The fundamental issue is the lack of regulatory oversight. Many of these tests exist in a grey area, carefully marketing themselves for “wellness” to avoid the rigorous validation required for true medical devices. This point is critical for understanding why the NHS cannot simply accept their findings. As one analysis highlights:
Many of these tests operate in a regulatory gray area by marketing themselves for ‘wellness’ or ‘ancestry’ purposes, not as medical devices. This protects them from the rigorous validation required for real diagnostic tools.
– Healthcare regulatory analysis, Analysis of commercial genetic testing regulatory frameworks
With over 80% of rare disorders having a genetic origin, the potential for genetic screening is enormous. However, a DTC test that reports a “slightly increased risk” for a condition can cause immense anxiety without providing a clear clinical path. The NHS prefers to use its own targeted, validated tests so that a positive result can be immediately followed by genetic counselling and a clear management plan. A DTC test provides data without a plan, often leaving the patient and their GP in a state of uncertainty.
Ultimately, paying for a private DNA test is a personal choice. But it should be an informed one. Treat it as a tool for personal curiosity, not as a shortcut to a medical diagnosis. The real, actionable medical insights will continue to come from the regulated, expert-led environment of the healthcare system.
Start today by organizing your medical history. A clear, structured record is the most powerful tool you can bring to your next consultation, whether it’s with a human doctor or an AI-assisted one. This proactive step transforms you from a passive patient into the active CEO of your own health.